 (a) Dimitri Mendeleev is widely credited with creating (b) the first periodic table of the elements. Although Mendeleev and Meyer had a long dispute over priority, Mendeleev’s contributions to the development of the periodic table are now more widely recognized (Figure 1).įigure 1. The discoveries of gallium (1875) and germanium (1886) provided great support for Mendeleev’s work. 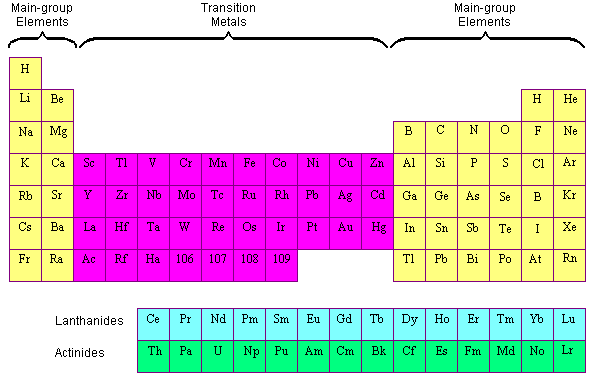
But Mendeleev went one step further than Meyer: He used his table to predict the existence of elements that would have the properties similar to aluminum and silicon, but were yet unknown. Both published tables with the elements arranged according to increasing atomic mass. Fluorine (F), chlorine (Cl), bromine (Br), and iodine (I) also exhibit similar properties to each other, but these properties are drastically different from those of any of the elements above.ĭimitri Mendeleev in Russia (1869) and Lothar Meyer in Germany (1870) independently recognized that there was a periodic relationship among the properties of the elements known at that time. For example: Li, Na, and K are much more reactive than are Ca, Sr, and Ba Li, Na, and K form compounds with oxygen in a ratio of two of their atoms to one oxygen atom, whereas Ca, Sr, and Ba form compounds with one of their atoms to one oxygen atom. However, the specific properties of these two groupings are notably different from each other. A second grouping includes calcium (Ca), strontium (Sr), and barium (Ba), which also are shiny, good conductors of heat and electricity, and have chemical properties in common. One such grouping includes lithium (Li), sodium (Na), and potassium (K): These elements all are shiny, conduct heat and electricity well, and have similar chemical properties. Identify metals, nonmetals, and metalloids by their properties and/or location on the periodic tableĪs early chemists worked to purify ores and discovered more elements, they realized that various elements could be grouped together by their similar chemical behaviors.Predict the general properties of elements based on their location within the periodic table.State the periodic law and explain the organization of elements in the periodic table.You will be able to download the images using the download button, whether you are using Windows, Mac, iOS, or Android. If you are using a mobile phone, you can also use the browser's menu drawer. Remember to bookmark Periodic Table Color Coded Metals Nonmetals Metalloids with Ctrl + D (PC) or Command + D (macOS). "Remember the worth of Souls is Great": ChemStdBlogpost Printable Periodic Table Metals Nonmetals Metalloids | Cabinets Matttroyīonds From Atoms - Stone Cold Chemistry Talk The Periodic Table | CHEM 1305 Introductory Chemistry Metals, Metalloids and Non-metals (By: Liliane Suarez)ġ.2) Atoms and the Periodic Table - Science with Mrs PizzimentiĬLILstore unit 2871: The classification of the elementsġ0 Difference Between Metals, Nonmetals And Metalloids (With Examples In addition to Periodic Table Color Coded Metals Nonmetals Metalloids above, here another images you might consider to download: Your students will love this activity! One way to classify elements in the periodic table is by metals, nonmetals, and metalloids.Ĭolor the Periodic Table (metals, nonmetals, metalloids, families) Activity The most reactive metal in the periodic table is francium. Copper is a reddish metal with the chemical symbol of cu. The metalloids and nonmetals, for example, don't fall neatly into the same column. Periodic Table Color Coded Metals Nonmetals Metalloids. 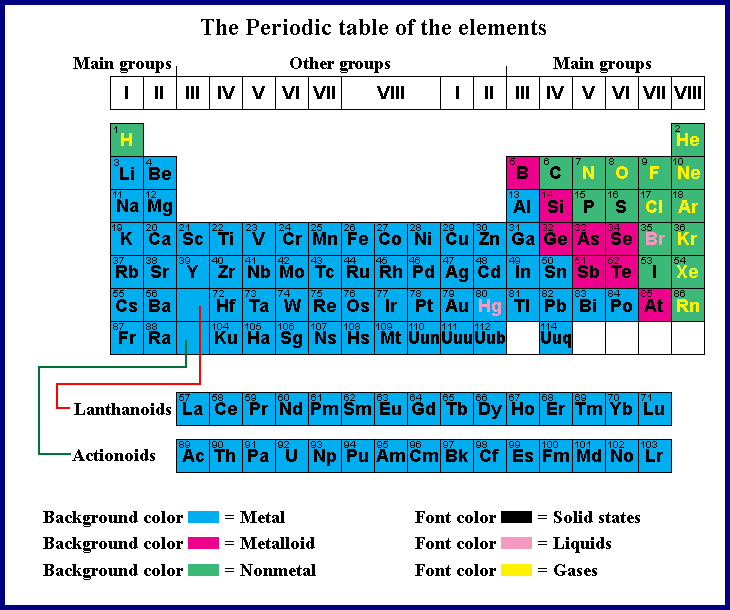 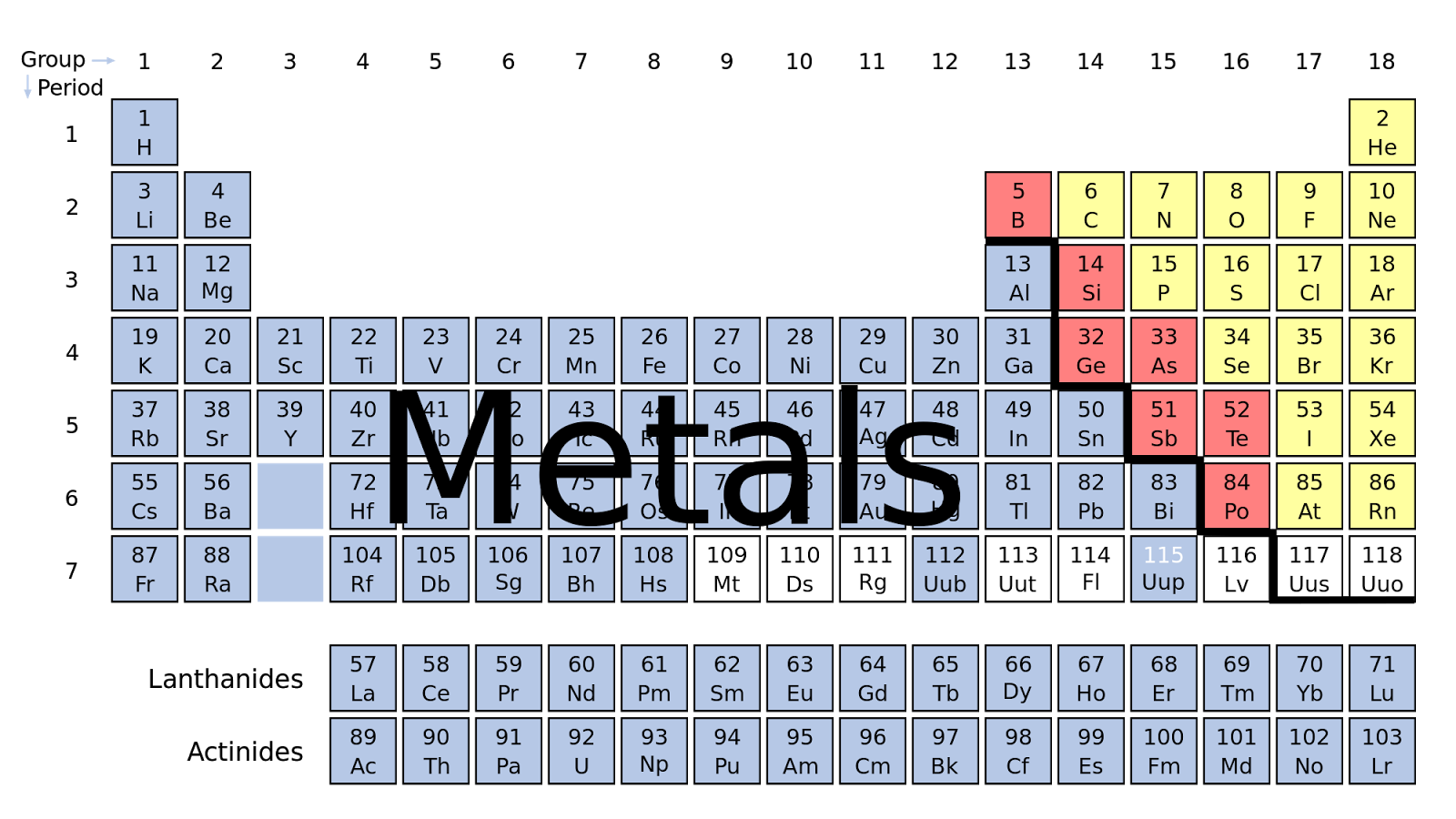
Periodic table color coded metals nonmetals metalloids worksheet periodic table color coded with key nonmetals metals and metalloids current periodic table color coded periodic table color coated periodic table coloring assignment chemistry periodic table periodic table groups periodic table labeled 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
